Collaborative work between a group of Takashi Toda and Masashi Yukawa, members of HiHA, and two groups in the Francis Crick Institute (Thomas Surrey and Martin R. Singleton) was published in Journal of Cell Science online.
This study has shown that two conserved microtubule associated proteins, Dis1/TOG and Mal3/EB, physically interact in a non-cannonical manner and play a critical role in high-fidelity chromosome segregation in fission yeast.
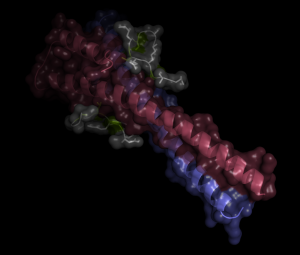
The image below illustrates a novel interaction mode between EB1 and XMAP215/TOG members in fission yeast. It is based upon the crystallographic data of a binding complex between Mal3/EB1 and a peptide corresponding to the C-terminal region of Dis1/TOG (21 amino acid residues corresponding to 833-853). Dis1 binds Mal3 in a novel, unconventional manner, in which the binding does not depend upon a canonical SXIP motif. Instead a short peptide (837AGSMLQKPTQFSRP849) binds Mal3, and L841, P844 and F847 (underlined). These three amino acids interact with a hydrophobic cavity formed within the Mal3 C-terminal region that typically interacts with the canonical SXIP motif (shown in this image).
Article
An unconventional interaction between Dis1/TOG and Mal3/EB1 promotes the fidelity of chromosome segregation
Yuzy Matsuo, Sebastian P. Maurer, Masashi Yukawa, Silva Zakian, Martin R. Singleton, Thomas Surrey and Takashi Toda
J Cell Sci 2016 : doi: 10.1242/jcs.197533
【Abstract】